|
3/16/2024 0 Comments Niels bohr atomic model
Bohr used the term energy levels (or shells) to describe these orbits of differing energy.The Bohr model shows that the electrons in atoms are in orbits of differing energy around the nucleus (think of planets orbiting around the sun). 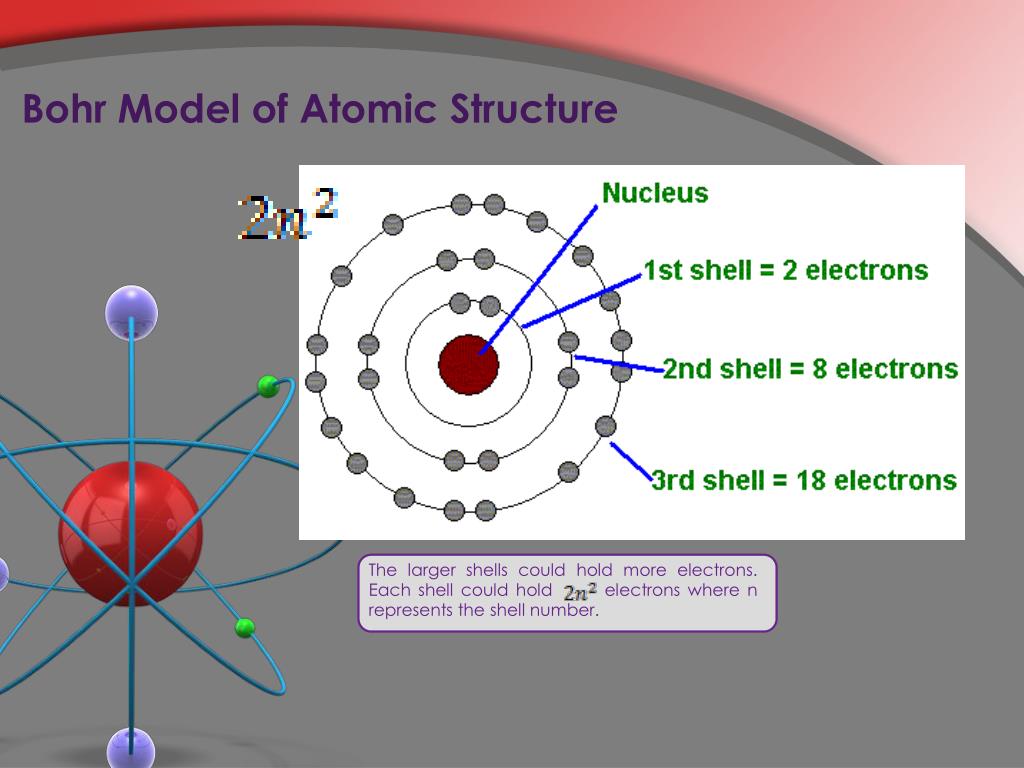
Niels Bohr, a Danish scientist, explained this line spectrum while developing a model for the atom: Bohr said that the electron had to release energy to change its energy so the differences between the energies of light seen in the atomic spectrum should correspond to the differences in energies of the energy levels. He used these energies to find the energies that the single electron in the hydrogen atom could have. Bohr found the energy of the colors of light that the hydrogen atom released. This led Bohr to theorize that electrons only have certain energies in an atom and they had to be in energy levels. Each color corresponds to a specific amount of energy however when the light given off by the hydrogen atom was passed through a prism, only certain colors of light could be seen. When white light is diffracted with a prism, all the colors of the visible spectrum can be seen. The Bohr model was based on his observations of the atomic emissions spectrum of the hydrogen atom. The Rutherford model had several flaws that the Bohr model overcame. This model was proposed in 1913 by Niels Bohr and was really an expansion on the Rutherford model of 1911. The energy difference between the initial and final orbit is emitted by the atom in bundles of electromagnetic radiation called photons. When an electron changes orbits, it does so in a sudden quantum leap. In this book we have a remarkable narrative of the genesis, application, reception, and use of one of the pivotal theories of modern physics.A simple definition of Bohr’s atomic model is: electrons orbit the nucleus at set distances. Kragh examines Bohr's atom on its own terms, not merely as a stepping-stone to later quantum theory. Usually exiled to introductory chapters in histories of quantum mechanics, this instantly recognizable model has long needed a focused investigation. "Around the world, the Bohr model of the atom is visually synonymous with "science." Despite its universality, the Bohr atom has waited a hundred years for Kragh's thorough study. 
Stuewer, History of Science and Technology, University of Minnesota We have here what promises to be the definitive history of the Bohr theory of the atom." It is unique in emphasizing and documenting that Bohr conceived his theory as a comprehensive theory of the constitution of matter, one that applied to both atoms and molecules, that is, to the domains of both physics and chemistry, whose practitioners responded to it in profoundly different ways. It explores the development of the revolutionary Bohr atom from its birth in 1913 and extensions during the next five years to its supersession by modern quantum mechanics in the mid 1920s, treating at each stage its successes and failures and its positive and negative receptions in Europe and the United States. 
"This compelling biography of a scientific theory, that of the Bohr atom, is unprecedented and unmatched in depth and breadth. Klaus Hentschel, Professor and Director of the History Department, Stuttgart University Written by an experienced historian of science, it surveys the full breadth of the literature on its history and implications in physics, chemistry, astrophysics and far beyond, including excerpts from many still unpublished sources such as letters or manuscripts. This book is likely to become the definitive history of the development of Bohr's atomic model. ContentsĢ: On the constitution of atoms and moleculesĩ: Appendix: The philosophers' atom Customer Reviews By discussing the theory in its entirety it becomes possible to understand why it developed as it did and thereby to use it as an example of the dynamics of scientific theories. Moreover, it discusses the theory as Bohr originally conceived it, namely, as an ambitious theory covering the structure of atoms as well as molecules. Contrary to most other accounts of the Bohr atom, the book presents it in a broader perspective which includes the reception among other scientists and the criticism launched against it by scientists of a more conservative inclination. At the same time it offers new insight into Bohr's peculiar way of thinking, what Einstein once called his 'unique instinct and tact'. Niels Bohr and the Quantum Atom is the first book that focuses in detail on the birth and development of Bohr's atomic theory and gives a comprehensive picture of it.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
